2Department of Cardiology, Kozluk State Hospital, Batman, Türkiye
3Department of Cardiology, Sincan Training and Research Hospital, Ankara, Türkiye
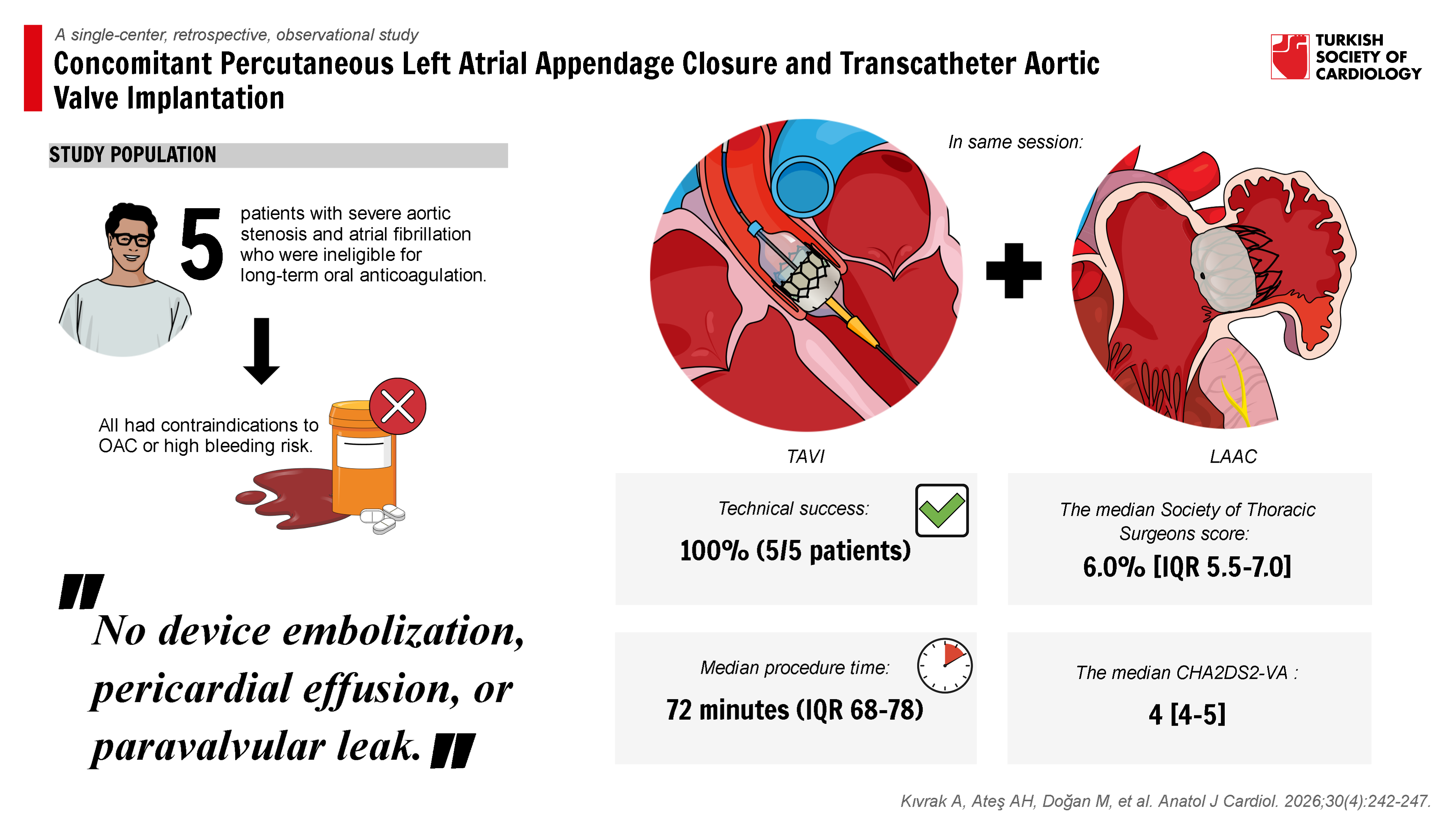
Abstract
Background: Patients with atrial fibrillation (AF) undergoing transcatheter aortic valve implantation (TAVI) often require long-term oral anticoagulation (OAC), which may not be appropriate for those at high bleeding risk. Performing left atrial appendage closure (LAAC) during TAVI can reduce the risk of thromboembolism while avoiding the need for prolonged anticoagulation.
Methods: This single-center study included 5 consecutive patients with severe aortic stenosis and AF who underwent same-session TAVI and LAAC between October 2024 and March 2025. All had contraindications to OAC or high bleeding risk. Procedural details and early outcomes were recorded. Technical success was defined according to Valve Academic Research Consortium-3 (VARC-3) (TAVI) and Munich/The Society for cardiovascular Angiography & Interventions (SCAI) and the Heart Rhythm Society criteria (LAAC). Continuous variables are presented as mean ± SD or median interquartile range (IQR), and categorical variables as n (%).
Results: Mean age was 75.6 ± 8.4 years; 40% were male. The median Society of Thoracic Surgeons score was 6.0% [IQR 5.5-7.0], median CHA2DS2-VA was 4 [4-5], and median hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile INR, elderly, drugs/alcohol concomitantly was 3 [3-4]. All patients received a Meril valve; LAAC devices included Amulet (n = 3) and LAmbre (n = 2). Technical success was achieved in all cases. There were no intra-periprocedural complications, major bleeding (Bleeding Academic Research Consortium ≥3), stroke/transient ischemic attack, or vascular complications. But Kidney Disease: Improving Global Outcomes stage 1 acute kidney injury was observed only in 1 (20%) patient. The median hospital stay was 4 [IQR 3-6] days.
Conclusion: In this study, same-session TAVI and LAAC in AF patients with high bleeding risk were technically feasible and showed an acceptable short-term safety profile. Larger, prospective studies with longer follow-up are needed to confirm these results.
Graphical Abstract
Highlights
- The combined transcatheter aortic valve implantation–left atrial appendage closure (TAVI-LAAC) procedure was technically successful in all 5 patients (100%) with no procedural complications, demonstrating the safety and feasibility of this approach.
- Median procedure time was 72 minutes with a contrast volume of 65 mL, indicating that the combined procedure can be performed efficiently without excessive procedural burden.
- No major adverse events, including stroke, major bleeding, or pericardial complications, occurred during the index hospitalization, supporting the early safety profile of simultaneous TAVI-LAAC.
- This combined approach may be particularly beneficial for high-risk elderly patients with atrial fibrillation requiring both TAVI and stroke prevention, potentially reducing the need for multiple procedures and prolonged anticoagulation.
Introduction
Severe aortic stenosis (AS) presents a significant procedural challenge, especially in elderly patients with atrial fibrillation (AF), a subgroup characterized by both thromboembolic and hemorrhagic risks.1,
Patients with AF who are poor candidates for OAC due to either prior major bleeding, frailty, or inability to tolerate therapy remain at high risk for stroke and vulnerable to hemorrhagic complications. LAAC offers a non-pharmacological alternative by eliminating the primary source of thrombus formation in non-valvular AF and is particularly useful in those with contraindications to OAC or a high bleeding risk (HAS-BLED score of ≥3).4 In TAVI candidates, who are often elderly and have comorbidities, the combined approach of LAAC and TAVI in a single session has demonstrated technical feasibility and safety in small case series, especially in OAC-ineligible patients.5
The concept of performing TAVI and LAAC in a single session aims to minimize procedural burden, reduce cumulative exposure to anesthesia and vascular access, and potentially shorten hospital stay. Several small case series and observational studies have reported high technical success rates with acceptable complication profiles, suggesting that the approach may be a viable option for selected high-risk patients.3,
Given the lack of data from real-world cohorts and the limited representation of OAC-ineligible patients in larger trials, further evidence is required to define the procedural feasibility and short-term safety of same-session TAVI and LAAC. In this study, a single-center experience with 5 high-risk AF patients who underwent combined TAVI and LAAC in a single session was reported. All patients had contraindications to long-term OAC due to major bleeding events or thromboembolic events occurring while on therapy. The primary objective of this study was to assess technical success, while secondary endpoints focused on in-hospital complications and early post-procedural outcomes.
Methods
Study Population
This was a single-center, retrospective, observational study including 5 consecutive patients with severe, symptomatic AS and coexisting AF who underwent combined TAVI and LAAC in a single procedural session between October 2024 and March 2025. All patients were ineligible for long-term OAC due to a history of major bleeding (e.g., gastrointestinal or ocular hemorrhage) or thromboembolic events occurring while on therapeutic OAC. The decision to perform combined TAVI and LAAC was made by the institutional Heart Team, based on clinical presentation and comorbidities. The study was conducted in accordance with the principles of the Declaration of Helsinki. The protocol was approved by the Hacettepe University Health Sciences Research Ethics Committee (Date: August 26, 2025; Decision number: 2025/16-61; Study registration number: SBA 25/746). Waived by the ethics committee for this retrospective analysis of anonymized medical records.
Pre-procedural Evaluation
All patients underwent a comprehensive pre-procedural assessment, including detailed clinical history, physical examination, laboratory testing, transthoracic echocardiography, and multislice computed tomography (CT) for annular sizing and vascular access evaluation. Transesophageal echocardiography (TEE) was performed to assess the morphology and dimensions of the left atrial appendage (LAA) and to exclude pre-existing thrombus. Echocardiographic parameters, including peak and mean transvalvular gradients and aortic valve area measurements, confirmed the severity of AS in accordance with current guidelines. Risk stratification was performed using the Society of Thoracic Surgeons (STS) score, CHA2DS2-VA, and HAS-BLED scores, calculated from baseline clinical and echocardiographic data.
Procedural Details
All procedures were performed under general anesthesia in a cardiac catheterization laboratory. Following sterile preparation and obtaining vascular access, transfemoral TAVI was performed using the balloon-expandable Myval transcatheter heart valve (Meril Life Sciences, Vapi, India), with device size selection based on pre-procedural multislice CT annular measurements. After successful valve implantation, TEE was introduced for LAAC planning. Venous access was obtained via the contralateral femoral vein, and transseptal puncture was performed at the inferoposterior aspect of the interatrial septum under fluoroscopic and TEE guidance. Device sizing and selection were based on TEE measurements of the LAA landing zone and ostium. Zero-contrast LAAC was performed using Amulet LAA Occluder (Abbott, Chicago, IL, USA) in 3 patients and LAmbre LAA Closure System (Lifetech Scientific, Shenzhen, China) in 2 patients. Devices were deployed under fluoroscopic and TEE visualization, ensuring optimal position and seal before release, as described in previous studies (
Study Endpoints
The primary endpoint was technical success, defined as successful deployment of both the transcatheter aortic valve and the LAAC device in the intended position, with no need for additional unplanned interventions and without in-hospital mortality. For the TAVI procedure, technical success was defined according to the Valve Academic Research Consortium-3 (VARC-3) criteria, which include the absence of procedural mortality, correct positioning of a single prosthetic heart valve into the proper anatomical location, and intended valve performance with no prosthesis-patient mismatch or significant paravalvular leak at hospital discharge.9 For the LAAC procedure, technical success was defined based on the Munich Consensus Document, which provides standardized definitions for procedural outcomes, endpoints, and data collection in clinical studies.10 These criteria require successful device deployment with complete LAAC or residual peri-device leak ≤ 5 mm, stable device position confirmed by imaging, and no device embolization or surgical intervention.
Secondary endpoints included the occurrence of vascular complications (major or minor) according to VARC-3, major bleeding events defined as Bleeding Academic Research Consortium (BARC) type ≥ 311 pericardial effusion or tamponade requiring intervention, stroke or transient ischemic attack (TIA), new permanent pacemaker implantation, and acute kidney injury (AKI) according to the Kidney Disease: Improving Global Outcomes (KDIGO) classification.
Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables were assessed for distribution using the Shapiro–Wilk test. Given the small sample size and non-normal distribution of most variables, continuous data are presented as median [interquartile range (IQR)], and categorical data are presented as counts and percentages. No imputation was performed for missing data. Descriptive statistics were used to summarize baseline characteristics, procedural details, and outcomes.
Results
The study included 5 patients with a mean age of 75.6 ± 8.4 years, of whom 2 (40.0%) were male. The mean body mass index was 24.45 ± 2.11 kg/m². Four patients (80.0%) had a history of coronary artery disease and hypertension, and 1 patient (20.0%) had diabetes mellitus. Non-paroxysmal AF was documented in 4 patients (80.0%). The median STS score was 6.0 (IQR 5.5-7.0). The median CHA2DS2-VA and HAS-BLED scores were 4 (IQR 4-5) and 3 (IQR 3-4), respectively. The primary indications for LAAC included gastrointestinal bleeding in 2 patients (40.0%), ischemic stroke despite OAC in 2 patients (40.0%), and systemic embolism (retinal artery occlusion) despite OAC in 1 patient (20.0%) (
All procedures were completed in a single session without intraprocedural complications. The median procedural time was 72 minutes (IQR 68-78), and the median contrast volume was 65 mL (IQR 60-70), reflecting the use of a zero-contrast strategy for LAAC with TEE and fluoroscopic guidance. All TAVI procedures were performed using balloon-expandable Myval valves (Meril Life Sciences, Gujarat, India), and LAAC was achieved with either Amulet (Abbott, Chicago, IL, USA) or LAmbre (Lifetech Scientific, Shenzhen, China) devices.
Postprocedural antithrombotic therapy at discharge involved clopidogrel alone in 2 patients (40.0%) due to prior gastrointestinal bleeding, and aspirin plus apixaban in the remaining 3 patients (60.0%) who had a history of thromboembolism despite OAC, as shown in
During the in-hospital period, no deaths, myocardial infarctions, or strokes were recorded. There were no cases of device embolization, pericardial effusion, or major vascular complications. One patient developed AKI, which was classified as stage 1 according to the KDIGO criteria, and was managed conservatively without the need for renal replacement therapy. No major or life-threatening bleeding events were observed, and there were no cases of clinically significant paravalvular leak after TAVI or residual peri-device leak following LAAC (
Discussion
Performing TAVI and LAAC simultaneously has become a viable treatment option for patients with AF and severe AS who are at high risk for repeated procedures. This combined approach may help reduce risks associated with multiple interventions, such as repeated anesthesia, longer hospital stays, vascular access issues, and increased contrast use. Previous studies, including the randomized study reported by Kapadia et al,6 have shown promising results in terms of procedural success and early safety.3,
An increasing amount of evidence shows that performing both TAVI and LAAC in a single session is feasible and can help avoid repeated anesthesia and vascular access, which is especially important in high-risk patients. In the authors’ experience, this combined approach was further supported by using a zero-contrast method for LAAC, relying on TEE and fluoroscopic guidance. This minimized renal risk while ensuring procedural safety. On the other hand, cardiovascular interventions using general anesthesia are generally safe, but cumulatively are associated with cognitive decline and neurodegeneration in the elderly. Perioperative brain health should be prioritized for older and vulnerable patients, particularly those who have multiple interventional procedures using anesthesia.13 These findings align with previous reports of technical success and positive early outcomes in similar populations.6,
Our findings support previous observational and randomized studies indicating that concomitant TAVI and LAAC are both feasible and safe in selected high-risk patients. The “One-Stop Shop” study showed comparable 30-day outcomes with combined versus isolated TAVI,3 while Kleinecke et al14 found no increase in complications when LAAC was performed with other structural interventions. More recently, the WATCH-TAVR trial confirmed that the combined approach is not inferior in terms of death, stroke, and major bleeding at 2 years.6 Collectively, this data support LAAC integration during TAVI as a viable alternative for AF patients with high bleeding risk or contraindications to OAC.
An essential part of this study is the use of a zero-contrast strategy during LAAC, which led to a relatively low median contrast volume compared to larger cohorts. In WATCH-TAVR, the excessive contrast used for the combined procedure was about 119 mL, significantly higher than the median 65 mL reported in the present cohort. Excessive contrast exposure is a known factor contributing to AKI after TAVI, especially in elderly patients with baseline renal dysfunction. Previous consensus statements have highlighted strategies for kidney protection, including reducing contrast and relying on echocardiographic guidance whenever possible.15 Based on the authors’ experience, only 1 patient developed stage 1 AKI and was managed conservatively, highlighting the potential benefit of a contrast-sparing approach in high-risk groups.
In this cohort, there were no major periprocedural complications such as device embolization, pericardial effusion, stroke, or major bleeding, which aligns with findings from prior series. The case series by Freire et al5 reported 7 patients undergoing concomitant TAVR and LAAC, all with successful implantation and no major adverse events during follow-up. Similarly, a meta-analysis including 482 patients indicated that combined procedures did not significantly raise the risk of stroke, bleeding, or death compared to isolated TAVR. However, a higher rate of vascular complications was observed.16 These results suggest that although the overall safety profile of simultaneous TAVI and LAAC is acceptable, managing vascular access remains crucial, especially considering the added procedural complexity and the common frailty in this patient group.
The best antithrombotic strategy after combined TAVI and LAAC remains uncertain. Current European Society of Cardiology guidelines recommend lifelong OAC for AF after TAVI, but the high bleeding risk in elderly and frail patients challenges this approach.17 By eliminating the need for long-term anticoagulation, LAAC provides a potential advantage in this subgroup. Kapadia et al6 demonstrated in the WATCH-TAVR trial that dual antithrombotic therapy after combined procedures offered acceptable thromboembolic protection and a lower bleeding risk compared with OAC. The present practice of using short-term dual antithrombotic treatment followed by single antiplatelet therapy aligns with this evidence. This indicates that a personalized antithrombotic regimen after combined procedures can be both safe and effective.
These findings contribute to the growing evidence supporting the feasibility of performing concomitant TAVI and LAAC in selected patients with severe AS and AF who are at high risk of bleeding. While previous studies have mostly included diverse patient populations or single-center experiences,3,
This study is limited by its single-center, retrospective design and relatively small sample size, which may restrict the generalizability of the findings. Additionally, the short follow-up duration prevents definitive conclusions about long-term outcomes, and the lack of a control group limits direct comparisons with isolated TAVI or LAAC procedures. Despite these limitations, these results support the feasibility and short-term safety of performing TAVI and LAAC together in carefully selected high-risk patients with severe AS and AF. These findings suggest that the combined approach could be a reasonable treatment option for patients with contraindications to long-term anticoagulation, warranting validation in larger multicenter studies with longer follow-up.
Footnotes
References
- Smith CR, Leon MB, Mack MJ. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187-2198.
- Achenbach S, Delgado V, Hausleiter J, Schoenhagen P, Min JK, Leipsic JA. SCCT expert consensus document on computed tomography imaging before transcatheter aortic valve implantation (TAVI)/transcatheter aortic valve replacement (TAVR). J Cardiovasc Comput Tomogr. 2012;6(6):366-380.
- Attinger-Toller A, Maisano F, Senn O. “One-Stop Shop”: safety of combining transcatheter aortic valve replacement and left atrial appendage occlusion. JACC Cardiovasc Interv. 2016;9(14):1487-1495.
- Reddy VY, Möbius-Winkler S, Miller MA. Left atrial appendage closure with the Watchman device in patients with a contraindication for oral anticoagulation: the ASAP study (ASA Plavix Feasibility Study With Watchman Left Atrial Appendage Closure Technology). J Am Coll Cardiol. 2013;61(25):2551-2556.
- Freire AFD, Filippini FB, Bignoto TC. Combined transcatheter aortic valve replacement and left atrial appendage occlusion in patients ineligible for oral anticoagulation: a case series. J Cardiol Cases. 2022;26(3):181-185.
- Kapadia SR, Krishnaswamy A, Whisenant B. Concomitant left atrial appendage occlusion and transcatheter aortic valve replacement among patients with atrial fibrillation. Circulation. 2024;149(10):734-743.
- Ates AH, Kivrak A, Karakulak UN. Should left atrial appendage closure be considered in resistant left atrial appendage thrombus cases? ‘Former Foe, New Ally’. Pacing Clin Electrophysiol. 2024;47(4):583-590.
- Kivrak A, Ates AH, Canpolat U. Management of device embolization following left atrial appendage closure: two cases and a review of the literature. J Arrhythm. 2025;41(4):e70139-.
- Généreux P, Piazza N, Alu MC. Valve Academic Research Consortium 3: Updated endpoint definitions for aortic valve clinical research. J Am Coll Cardiol. 2021;77(21):2717-2746.
- Tzikas A, Holmes DR, Gafoor S. Percutaneous left atrial appendage occlusion: the Munich consensus document on definitions, endpoints, and data collection requirements for clinical studies. Europace. 2017;19(1):4-15.
- Mehran R, Rao SV, Bhatt DL. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123(23):2736-2747.
- Taramasso M, Maisano F, Nietlispach F. TAVI and concomitant procedures: from PCI to LAA closure. EuroIntervention. 2015;11(Suppl W):W96-W100.
- Taylor J, Robledo KP, Medel V. Association between surgical admissions, cognition, and neurodegeneration in older people: a population-based study from the UK Biobank. Lancet Healthy Longev. 2024;5(9):100623-.
- Kleinecke C, Buffle E, Link J. Amplatzer left atrial appendage closure: single versus combined procedures. Catheter Cardiovasc Interv. 2021;97(7):E973-E981.
- Barbanti M, Gargiulo G, Tamburino C. Renal dysfunction and transcatheter aortic valve implantation outcomes. Expert Rev Cardiovasc Ther. 2016;14(12):1315-1323.
- Ahmed H, Ismayl M, Palicherla A. Outcomes of simultaneous left atrial appendage closure in atrial fibrillation patients undergoing transcatheter aortic valve replacement. Ann Med Surg (Lond). 2024;86(8):4316-4318.
- Van Gelder IC, Rienstra M, Bunting KV. ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS) Developed by the task force for the management of atrial fibrillation of the European Society of Cardiology (ESC), with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Endorsed by the European Stroke Organisation (ESO). Eur Heart J. 2024;2024():ehae176-.


