Abstract
Background: Transcatheter aortic valve replacement (TAVR) is the standard therapy for severe aortic stenosis, particularly in elderly patients with comorbidities. Simple biomarkers to predict midterm mortality are still needed. This study evaluated the prognostic value of the preprocedural neutrophil percentage-to-albumin ratio (NPAR) for 2-year all-cause mortality after TAVR.
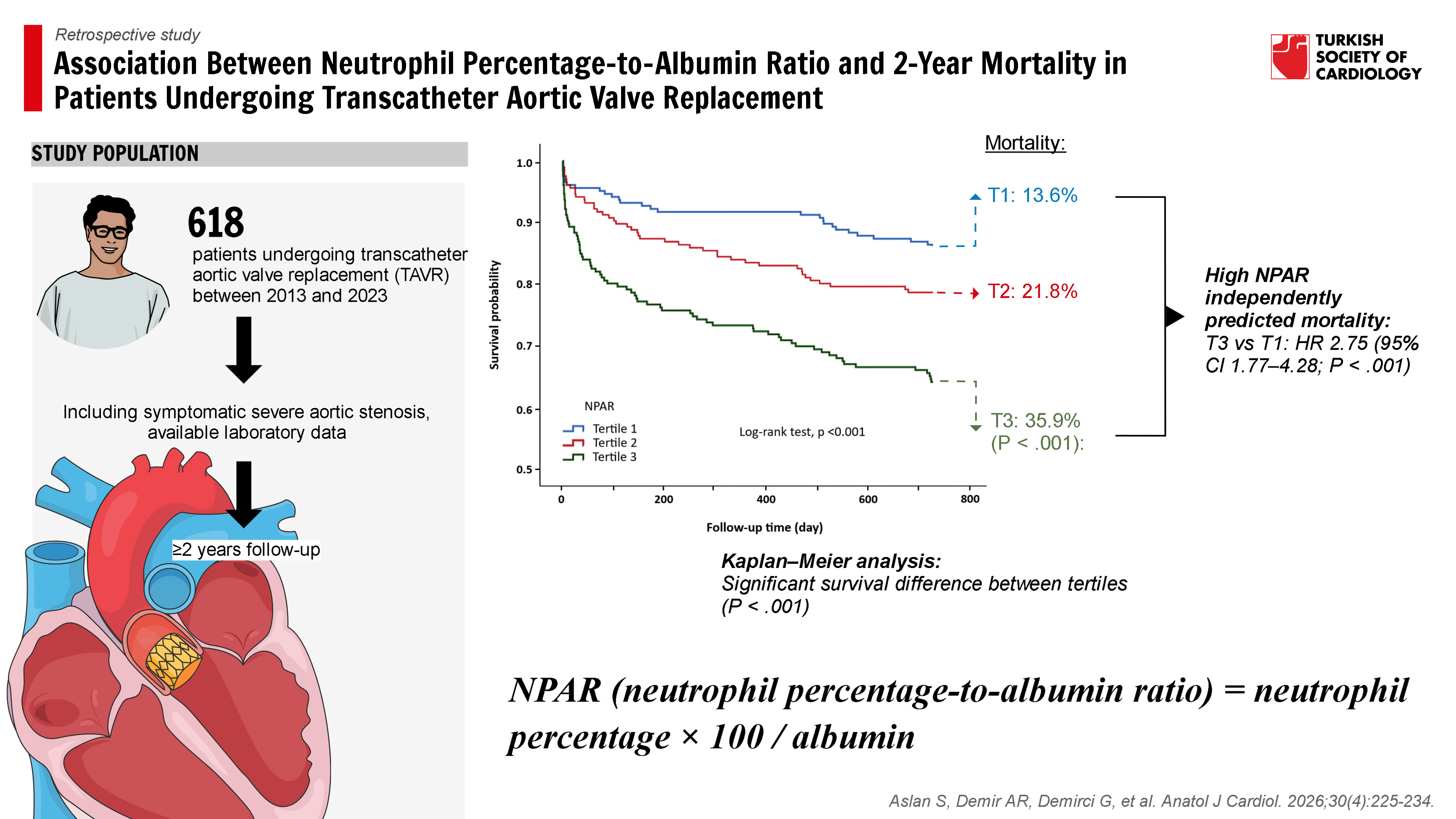
Methods: A total of 618 patients undergoing TAVR between 2013 and 2023 were retrospectively analyzed. NPAR was calculated as neutrophil percentage × 100 / albumin (g/dL), and patients were classified into tertiles. The prognostic role of NPAR was assessed using Cox regression, Kaplan–Meier survival analysis, and receiver operating characteristic curves.
Results: Baseline characteristics were similar across tertiles, but higher NPAR was associated with elevated inflammation and lower albumin levels. In multivariable Cox analysis, high NPAR independently predicted 2-year mortality (T3 vs. T1: hazard ratio [HR] 2.75, 95% CI 1.77-4.28; P < .001). In a model including both categorical NPAR and Society of Thoracic Surgeons Predicted Risk of Mortality (STS-PROM), tertile 3 of NPAR (HR 2.49,
95% CI 1.61-3.85; P < .001) and STS-PROM (HR 1.31, 95% CI 1.12-1.53; P = .001) remained independent predictors, indicating incremental prognostic value of NPAR beyond established surgical risk scores. Kaplan–Meier curves showed the lowest survival in the highest tertile (35.9% mortality at 2 years). Receiver operating characteristic analysis confirmed
NPAR had the best discriminatory ability (area under the curve = 0.703).
Conclusion: Preprocedural NPAR is an independent, low-cost, and readily available biomarker for predicting midterm mortality after TAVR. Its integration into risk models may improve prediction accuracy and help guide patient management.
Graphical Abstract
Highlights
- Neutrophil percentage-to-albumin ratio (NPAR) provides a simple and low-cost biomarker for risk stratification in transcatheter aortic valve replacement (TAVR) patients.
- Higher NPAR levels are independently associated with increased all-cause mortality over 2 years.
- This study is the first to demonstrate the prognostic utility of NPAR in the TAVR population.
- Neutrophil percentage-to-albumin ratio offers incremental prognostic value beyond STS-PROM, supporting its integration into existing risk models.
Introduction
Transcatheter aortic valve replacement (TAVR) is increasingly being used as an alternative to surgery in patients with severe symptomatic aortic stenosis. While current guidelines recommend TAVR for intermediate- and high-risk patients, studies demonstrating similar efficacy to surgery in low-risk groups have made it applicable to all risk groups.1-
In recent years, hematological and biochemical markers reflecting the relationship between systemic inflammation, nutritional status, and mortality have attracted attention.10-
Therefore, this study aimed to investigate the relationship between NPAR and 2-year all-cause mortality, and to evaluate the clinical utility of NPAR in predicting mortality after TAVR.
Methods
Study Design and Population
A retrospective analysis was performed on consecutive patients who underwent TAVR at this institution, a tertiary cardiac center, from February 2013 to June 2023. The study was conducted in accordance with the Declaration of Helsinki and approved by the Local Ethics Committee (Date: August 19, 2025; Decision No.: 2025.07-75). Inclusion criteria were as follows: (1) TAVR was performed with a diagnosis of symptomatic severe aortic stenosis, (2) preprocedural complete blood count (CBC) and biochemistry data were available, and (3) at least 2 years of follow-up data were available. Exclusion criteria included active infection, malignancy, autoimmune disease, hematological malignancy, or use of immunosuppressive therapy, chronic liver disease, and patients referred to another center during follow-up or missing mortality data. Finally, a total of 618 eligible patients were included in the study (
Data Collection and Definitions
Study data were collected retrospectively through the hospital information system and patient follow-up files. Demographic characteristics (age, gender), comorbidities (hypertension, diabetes mellitus, coronary artery disease, peripheral artery disease, atrial fibrillation, stroke history), echocardiographic parameters (left ventricular ejection fraction [LVEF], aortic valve area, systolic pulmonary artery pressure [sPAP]), and preprocedure laboratory data (CBC, biochemistry, inflammatory markers) were systematically recorded.
Laboratory Analysis and Neutrophil Percentage-to-Albumin Ratio Calculation
All blood samples were collected within 24 hours before the TAVR procedure. Neutrophil percentage and serum albumin levels were measured from the same sample. Neutrophil percentage-to-albumin ratio, the ratio of these 2 variables, was calculated using the following formula: NPAR = Neutrophil percentage (%) × 100 / Albumin (g/dL). Neutrophil percentage-to-albumin ratio values were divided into 3 tertiles for use in statistical analyses in the study: low NPAR (Tertile 1), medium NPAR (Tertile 2), and high NPAR (Tertile 3).
Transcatheter Aortic Valve Replacement Procedure
All patients were thoroughly evaluated by a multidisciplinary cardiac team and considered candidates for TAVR after being determined to be at high risk for valve surgery. All TAVR procedures were performed in a fully equipped hybrid operating room using a transfemoral approach. The method of anesthesia (local or general) was made at the discretion of the operator and the anesthesiologist, considering the clinical indications. Valve type and size were determined according to manufacturer recommendations based on computed tomography and echocardiography findings. The following transcatheter valve designs were used: CoreValve Evolut R (Medtronic, Minneapolis, Minn, USA), Portico (St. Jude Medical, St. Paul, Minneapolis, Minn, USA), Acurate neo2 (Boston Scientific, Marlborough, MA, USA), Sapien XT/Sapien 3 (Edwards Lifesciences, Irvine, California, USA), and Myval (Meril Life Sciences Private Ltd., Gujarat, India). Predilation of the native aortic valve was performed at the operator’s discretion. Postdilation under rapid pacing was considered in cases of moderate or severe paravalvular aortic regurgitation and/or underdilatation of the prosthesis. A percutaneous closure system (Perclose ProGlide; Abbott Laboratories, Abbott Park, Illinois) was used to close the vascular access site. A temporary pacemaker was placed as a backup for high-degree atrioventricular (AV) block when necessary. Postprocedural care was conducted in accordance with current guidelines.1,
Follow-Up and Clinical Endpoints
Patients were followed up at outpatient clinic visits and by telephone when necessary. Death information was verified with the National Death Notification System and hospital records. All clinical endpoints were defined according to Valve Academic Research Consortium-3 (VARC-3) criteria.16 The primary endpoint was 2-year all-cause mortality after TAVR. Secondary endpoints included 30-day stroke, major vascular complications, bleeding, acute kidney injury, myocardial infarction, and new permanent pacemaker implantation.
Statistical Analysis
All statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) version 24 software package (SPSS Inc., Chicago, Illinois, USA). The normality of distribution of continuous variables was evaluated using both graphical (histograms) and numerical methods. Among numerical tests, both Kolmogorov–Smirnov and Shapiro–Wilk tests were performed to ensure robustness. As each group included more than 200 patients, the Kolmogorov–Smirnov test was considered more appropriate for evaluating normality. One-way ANOVA was used for normally distributed data, and the Kruskal–Wallis test was used for non-normally distributed data. Comparisons between categorical data were made using the Chi-square or Fisher’s exact test. Continuous variables are presented as mean ± SD or median and interquartile range. Categorical variables are expressed as numbers (percentages). The predictive power of NPAR, neutrophil percentage, and albumin levels for mortality was assessed using receiver operating characteristic (ROC) curve analysis. The area under the curve (AUC) for each parameter was calculated and reported with its 95% CI. The optimal cut-off value of NPAR for predicting 2-year all-cause mortality was determined using the Youden index. Comparisons between ROC curves were performed using the DeLong test. All-cause survival time was analyzed using the Kaplan–Meier method, and survival curves were plotted for the 3 NPAR groups. The difference between the groups was assessed using the log-rank test. Median survival time and event incidence rates were calculated separately for 30 days, 1 year, and 2 years. Cox proportional hazards regression analyses were performed to identify independent risk factors associated with 2-year mortality. Analyses were performed as follows: Univariable analysis for all available variables; Multivariable Model 1, including demographic, clinical, and laboratory variables with
Results
Demographic and Clinical Characteristics
A total of 618 patients were included in this study. Demographic characteristics, comorbidities, laboratory, and echocardiographic parameters were compared among the 3 tertiles established according to NPAR (
Procedural Characteristics and Clinical Outcomes
A total of 66.8% of patients underwent TAVR under conscious sedation, and these rates were similar between the groups. There were no significant differences in procedure time, contrast amount, or the intensive care unit stay (
Predictors of Mortality
In the multivariable Cox proportional hazards regression analysis, 2 separate models were constructed (
Survival Analysis
Kaplan–Meier survival curves for 2-year all-cause mortality showed a significant difference in survival between NPAR tertiles (
Receiver Operating Characteristic Curve Analysis
The predictive power of NPAR, albumin, and neutrophil percentage for all-cause mortality was evaluated using ROC analyses. The ROC curves of the 3 variables are presented together in
Discussion
This is the first study to evaluate the relationship between preprocedural NPAR and all-cause mortality in patients undergoing TAVR. Mortality rates were observed to gradually increase with increasing NPAR values. Receiver operating characteristic curve analysis demonstrated that NPAR had a higher predictive value than albumin levels and neutrophil percentage. Multivariable Cox regression analysis demonstrated that elevated NPAR was independently associated with mortality. These results suggest that NPAR can be used as a simple, inexpensive, and accessible prognostic marker in TAVR patients.
Neutrophil percentage is one of the main cellular components involved in the acute phase response to inflammation. Neutrophils are known to play a central role in the triggering of cardiovascular events and contribute to endothelial dysfunction and atherothrombotic processes.17-
The patient group undergoing TAVR generally consists of individuals with advanced age, a high comorbidity burden, and increased frailty. In this patient group, postoperative outcomes and prognosis are closely related not only to the correction of valvular pathology but also to parameters such as systemic inflammatory burden and nutritional status. Previous studies have investigated the prognostic role of other inflammation-based indices in TAVR populations. For example, both the neutrophil-to-lymphocyte ratio (NLR) and the platelet-to-lymphocyte ratio have been associated with increased mortality and adverse cardiovascular events after TAVR, suggesting that systemic inflammation plays a central role in determining outcomes.10,
It is important to distinguish NPAR from the NAR. While NAR uses the absolute neutrophil count, NPAR is derived from neutrophil percentage in combination with serum albumin.28 This methodological difference may influence its prognostic implications, as percentage-based indices may better capture relative leukocyte distribution in systemic inflammation. To date, no study has evaluated NPAR in TAVR patients, making this work the first to address this gap in the literature. Importantly, in a dedicated model including both STS-PROM and NPAR, each variable remained independently associated with mortality, suggesting that NPAR provides incremental prognostic information beyond established surgical risk scores.
This study showed that as NPAR levels increased, 30-day, 1-year, and 2-year mortality rates increased significantly. Furthermore, Kaplan–Meier survival analysis revealed significantly shorter survival in the higher NPAR tertile. These findings suggest that NPAR is a strong predictor of clinical outcomes after TAVR. Kaplan–Meier survival analysis further confirmed that high NPAR levels were associated with significantly increased 2-year all-cause mortality. The approximately 22% difference in mortality rates across tertiles and the median survival time exceeding 130 days suggest that NPAR is a strong predictor of clinical outcome after TAVR.
In this study, patients with elevated NPAR experienced longer hospital stays following TAVR. This observation may reflect underlying biological mechanisms linking inflammation and frailty to adverse perioperative outcomes. Elevated systemic inflammation can impair wound healing and increase vulnerability to complications, while low albumin levels may signal impaired nutritional reserve and reduced physiological resilience.23-
In the ROC analysis, the AUC value for NPAR (0.703) indicates moderate predictive power, which is higher than its individual components, such as albumin (0.668) and neutrophil percentage (0.634). This finding suggests that composite markers such as NPAR may have stronger prognostic capacity than individual laboratory parameters, consistent with previous reports in patients with coronary artery disease and heart failure.13,
In multivariable Cox regression analysis, elevated NPAR remained an independent predictor of 2-year all-cause mortality, even after adjusting for classical risk factors such as age, LVEF, creatinine, and peripheral artery disease. Patients in the Tertile 3 group, in particular, had a 2.75-fold higher risk of mortality compared to the reference group (HR: 2.75; 95% CI: 1.77-4.28;
Risk assessment in TAVR patients has traditionally relied on scores such as STS-PROM and EuroSCORE II, developed for surgical populations and currently widely used.31-
Study Limitations
This study has several limitations. First, due to its retrospective design, a causal relationship cannot be established. Second, it was conducted at a single center with a limited patient population, which may affect the generalizability of the results. In addition, the long inclusion period (2013-2023) coincided with significant advances in TAVR technology and practice that could have influenced outcomes. Moreover, other markers of inflammation [e.g., CRP, interleukin (IL)-6, tumor necrosis factor-α (TNF-α)] were not included, preventing a comprehensive evaluation of inflammatory processes. Finally, patients with malignancy or autoimmune disease were excluded, although they constituted only a relatively small subgroup. This exclusion was necessary to minimize potential confounding effects of systemic inflammation or cachexia on NPAR values and is therefore unlikely to have significantly impacted the overall findings. Nevertheless, the large patient number, mid-term follow-up period, and adjustment for numerous potential confounding factors represent important strengths of this study.
Future Directions
Future studies should aim to further evaluate the prognostic value of NPAR and support its integration into clinical decision-making. In particular, temporal changes in NPAR should be monitored, and their association with short- and mid-term outcomes after TAVR should be clarified. Moreover, randomized controlled trials assessing the impact of preoperative interventions targeting inflammation and nutritional optimization on survival in patients with high NPAR levels are warranted. Combining NPAR with existing risk scoring systems to develop novel prognostic models could further enhance individualized patient management.
In conclusion, this study demonstrates that NPAR is an independent, accessible, and low-cost biomarker for predicting mid-term all-cause mortality in patients undergoing TAVR. Incorporating NPAR into routine clinical assessment could help refine risk stratification and guide postprocedural management in this growing patient population.
Footnotes
Artificial intelligence assisted technologies, such as large language models, chatbots, or image creators, were not used in the production of submitted work in this study.
References
- Vahanian A, Beyersdorf F, Praz F. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2022;43(7):561-632.
- Otto CM, Nishimura RA, Bonow RO. ACC/AHA guideline for the management of patients with valvular heart disease: a report of the American. J Am Coll Cardiol. 2020;77(4):e25-e197.
- Toff WD, Hildick-Smith D, Kovac J. Effect of transcatheter aortic valve implantation vs surgical aortic valve replacement on all-cause mortality in patients with aortic stenosis: a randomized clinical trial. JAMA. 2022;327(19):1875-1887.
- Popma JJ, Deeb GM, Yakubov SJ. Transcatheter aortic-valve replacement with a self-expanding valve in low-risk patients. N Engl J Med. 2019;380(18):1706-1715.
- Blankenberg S, Seiffert M, Vonthein R. Transcatheter or surgical treatment of aortic-valve stenosis. N Engl J Med. 2024;390(17):1572-1583.
- Mack MJ, Leon MB, Thourani VH. Transcatheter aortic-valve replacement in low-risk patients at five years. N Engl J Med. 2023;389(21):1949-1960.
- Van Mieghem NM, Deeb GM, Søndergaard L. Self-expanding transcatheter vs surgical aortic valve replacement in intermediate-risk patients: 5-year outcomes of the SURTAVI randomized clinical trial. JAMA Cardiol. 2022;7(10):1000-1008.
- Yamashita Y, Baudo M, Sicouri S. Effect of elevated body mass index on outcomes of transcatheter aortic valve replacement for severe aortic stenosis. Anatol J Cardiol. 2024;28(12):565-574.
- Kılıç R, Güzel T, Aktan A. Comparison of evolut-R 34 mm valve and smaller evolut-R valves in patients undergoing transcatheter aortic valve implantation and determination of mild paravalvular leak predictors. Anatol J Cardiol. 2024;28(2):109-117.
- Abushouk A, Agrawal A, Hariri E. Association between haematological parameters and outcomes following transcatheter aortic valve implantation at mid-term follow-up. Open Heart. 2022;9(2):e002108-.
- Seoudy H, Shamekhi J, Voigtländer L. C-reactive protein to albumin ratio in patients undergoing transcatheter aortic valve replacement. Mayo Clin Proc. 2022;97(5):931-940.
- Stan A, Călburean PA, Drinkal RK. Inflammatory status assessment by machine learning techniques to predict outcomes in patients with symptomatic aortic stenosis treated by transcatheter aortic valve replacement. Diagnostics (Basel). 2023;13(18):2907-.
- Cui H, Ding X, Li W, Chen H, Li H. The neutrophil percentage to albumin ratio as a new predictor of in-hospital mortality in patients with ST-segment elevation myocardial infarction. Med Sci Monit. 2019;25():7845-7852.
- Yu Y, Liu Y, Ling X. The neutrophil percentage-to-albumin ratio as a new predictor of all-cause mortality in patients with cardiogenic shock. BioMed Res Int. 2020;2020():7458451-.
- Wang X, Zhang Y, Wang Y. The neutrophil percentage-to-albumin ratio is associated with all-cause mortality in patients with chronic heart failure. BMC Cardiovasc Disord. 2023;23(1):568-.
- Généreux P, Piazza N, Alu MC. Valve academic research consortium 3: updated endpoint definitions for aortic valve clinical research. J Am Coll Cardiol. 2021;77(21):2717-2746.
- Della Bona R, Cardillo MT, Leo M. Polymorphonuclear neutrophils and instability of the atherosclerotic plaque: a causative role?. Inflamm Res. 2013;62(6):537-550.
- Carbone F, Mach F, Montecucco F. Update on the role of neutrophils in atherosclerotic plaque vulnerability. Curr Drug Targets. 2015;16(4):321-333.
- Bonaventura A, Montecucco F, Dallegri F. Novel findings in neutrophil biology and their impact on cardiovascular disease. Cardiovasc Res. 2019;115(8):1266-1285.
- Pende A, Artom N, Bertolotto M, Montecucco F, Dallegri F. Role of neutrophils in atherogenesis: an update. Eur J Clin Investig. 2016;46(3):252-263.
- Lin Y, Lin Y, Yue J, Zou Q. The neutrophil percentage-to-albumin ratio is associated with all-cause mortality in critically ill patients with acute myocardial infarction. BMC Cardiovasc Disord. 2022;22(1):115-.
- Cai C, Zhang B, Sun T. Neutrophil percentage to albumin ratio was associated with clinical outcomes in coronary care unit patients. Rev Cardiovasc Med. 2022;23(10):333-.
- Plakht Y, Gilutz H, Shiyovich A. Decreased admission serum albumin level is an independent predictor of long-term mortality in hospital survivors of acute myocardial infarction. Soroka acute myocardial infarction II (SAMI-II) project. Int J Cardiol. 2016;219():20-24.
- Wada H, Dohi T, Miyauchi K. Impact of serum albumin levels on long-term outcomes in patients undergoing percutaneous coronary intervention. Heart Vessels. 2017;32(9):1085-1092.
- Cheng CW, Lee CW, Chien SC, Yeh HI, Chen CY. Serum albumin was associated with a long term cardiovascular mortality among elderly patients with stable coronary artery disease. Acta Cardiol Sin. 2024;40(1):87-96.
- Shahim B, Redfors B, Lindman BR. Neutrophil-to-lymphocyte ratios in patients undergoing aortic valve replacement: the PARTNER trials and registries. J Am Heart Assoc. 2022;11(11):e024091-.
- Avci Y, Demir AR, Duran M. The prognostic value of C-reactive protein to albumin ratio in patients undergoing transcatheter aortic valve implantation. Acta Cardiol. 2022;77(10):930-936.
- Peng Y, Xue Y, Wang J. Association between neutrophil-to-albumin ratio and mortality in patients with cardiogenic shock: a retrospective cohort study. BMJ Open. 2020;10(10):e039860-.
- Yin R, Zhu W, Chen W, Shen J, Wu Y, Wang Z. The relationship between neutrophil percentage-to-albumin ratio and slow and normal coronary flow phenomenon. BMC Cardiovasc Disord. 2025;25(1):64-.
- Nair S, Ha FJ, Baradi A. The use of the neutrophil-lymphocyte ratio and platelet-lymphocyte ratio in predicting transcatheter aortic valve implantation mortality. Heart Lung Circ. 2024;33(12):1680-1687.
- Shahian DM, Jacobs JP, Badhwar V. The society of thoracic surgeons 2018 adult cardiac surgery risk models: Part 1-background, design considerations, and model development. Ann Thorac Surg. 2018;105(5):1411-1418.
- O’Brien SM, Feng L, He X. Adult cardiac surgery risk models: Part 2-Statistical Methods and Results. Ann Thorac Surg. 2018;105(5):1419-1428.
- Nashef SA, Roques F, Sharples LD. EuroSCORE II. Eur J Cardiothorac Surg. 2012;41(4):734-745.
- Condado JF, Junpaparp P, Binongo JN. Neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) can risk stratify patients in transcatheter aortic-valve replacement (TAVR). Int J Cardiol. 2016;223():444-449.
- Karaca M, Gumusdag A. Prognostic role of neutrophil percentage-to-albumin ratio in patients with non-ST-elevation myocardial infarction. Medicina (Kaunas). 2024;60(12):2101-.
- Sun T, Shen H, Guo Q. Association between neutrophil percentage-to-albumin ratio and all-cause mortality in critically ill patients with coronary artery disease. BioMed Res Int. 2020;2020():8137576-.


