2Department of Cardiology, Adana City Training and Research Hospital, Adana, Türkiye
3Department of Cardiology, Koşuyolu Heart Training and Research Hospital, İstanbul, Türkiye
Abstract
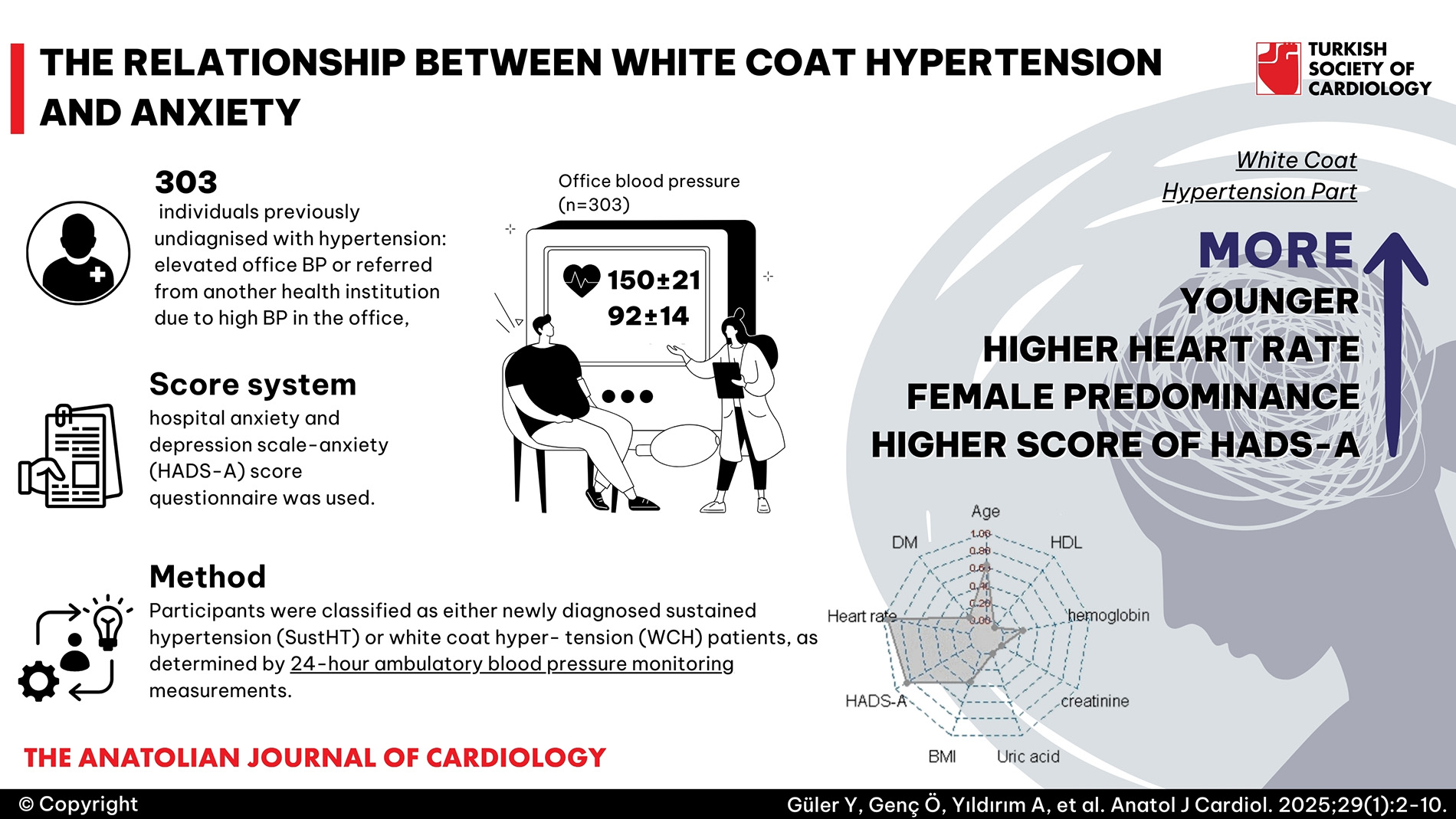
Background: To investigate the relationship between anxiety and white coat hypertension (WCH) using the hospital anxiety and depression scale-anxiety (HADS-A) score.
Methods: Participants lacking a pre-existing diagnosis of hypertension but displaying increased office blood pressure were included in this study. Subsequently, they were classified as either newly diagnosed sustained hypertension (SustHT) or white coat hypertension (WCH) patients, as determined by 24-hour ambulatory blood pressure monitoring measurements. The assessment of their anxiety levels was conducted using the HADS-A questionnaire. We performed regression, comparative, and sensitivity analyses to elucidate the association between anxiety and WCH.
Results: In this cohort of 303 consecutive individuals (mean age 54 years, 46% female), 81 (26.7%) patients were diagnosed with WCH. Those with WCH were younger (49 vs. 56 years, P < .001), had higher heart rate (85 vs. 76 bpm, P < .001) and exhibited a female predominance (56% vs. 43%, P = .049) compared to individuals with SustHT. The HADS-A was higher in WCH than in SustHT (9.0 ± 2.9 vs. 6.6 ± 2.6, P < .001). Furthermore, HADS-A showed positive correlation with systolic and diastolic pressures measured in the out-patient clinic (r = 0.523 and r = 0.387, respectively; P < .001 for both). The full model with HADS-A had better discriminatory power (Harrell’s c-index 0.82 vs. 0.77, P = .0025), increased calibration, and a greater net benefit than the base model without. The ROC curve analysis, using a cut-off of >6 for HADS-A, demonstrated a sensitivity of 76.5% and specificity of 53.6% in detecting WCH (Area Under the Curve = 0.72, P < .001).
Conclusions: Our study revealed that individuals with WCH, in comparison to those with SustHT, exhibit a higher level of anxiety as indicated by HADS-A.
Graphical Abstract
Highlights
- Anxiety and various types of psychological stress are thought to be major contributors to WCH. Yet, the link between WCH and anxiety is still debated. For some patients, a physician’s white coat and the atmosphere of their office can hold substantial emotional significance during follow-up visits, potentially acting as stimuli that contribute to increased anxiety.
- Despite the availability of numerous questionnaires designed to assess anxiety, there is a distinct lack of research examining the connection between WCH and anxiety using validated psychometric tools. The hospital anxiety and depression scale (HADS) is a widely used, easily administered, and highly effective tool that medical professionals frequently rely on to gauge patients’ anxiety and depression levels.
- In this study, we aimed to evaluate the relationship between HADS-Anxiety scores and WCH.
Introduction
White coat hypertension (WCH), commonly referred to as “office hypertension,” is characterized by elevated clinic blood pressure (BP) but normal ambulatory or home BP in untreated individuals.1,
Anxiety and other forms of psychological distress are believed to play a significant role in WCH.8-
While numerous questionnaires exist to quantify anxiety levels, there is a notable scarcity of studies investigating the relationship between WCH and anxiety using validated psychometric measures. The hospital anxiety and depression scale (HADS), developed by Zigmond and Snaith in 1983, has been translated and extensively utilized in numerous countries.15,
Methods
Patients, Definitions, and Study Design
A total of 315 patients, previously undiagnosed with hypertension and without prior antihypertensive treatment or medication for any other heart condition, yet exhibiting persistently elevated office BP or referred from another health institution due to high BP in the office, were consecutively enrolled in this single-center study conducted during routine cardiology examinations.
Individuals were excluded if they met any of the following criteria: (i) presence of depression, anxiety, or other psychiatric conditions (n = 3), (ii) secondary hypertension (n = 2), (iii) severe acute health conditions and/or previous cardiovascular morbid events (n = 3), and if they were taking antidepressants, anxiolytics, or any treatment known to increase BP or induce anxiety/depression (n = 4). Subsequently, 303 patients remained for the final analysis. The patients were categorized into 2 groups, WCH and SustHT. Each participant underwent a 12-lead electrocardiogram, clinical examination, and echocardiographic evaluation. For the diagnosis of WCH, office blood pressure measurements during at least 3 outpatient clinic visits were recorded as follows, in accordance with the guidelines from the European Society of Cardiology: Three BP measurements should be taken at intervals of at least 5 minutes, and additional measurements should be obtained only if there is a difference of more than 10 mm Hg between the first 2 measurements. Blood pressure is documented as the average of the last 2 blood pressure readings.4 Hypertension was defined as systolic BP of ≥140 mm Hg and/or diastolic BP of ≥90 mm Hg in the clinical setting. All participants underwent 24-hour ambulatory blood pressure monitoring (ABPM). They were categorized as newly diagnosed SustHT and WCH patients based on ABPM measurements. White coat hypertension was defined as the untreated state where office BP readings fall within the hypertensive range while ABPM values remain normal. On average, ABPM values are lower than office BP values, and the diagnostic threshold for hypertension is ≥130/80 mm Hg over 24 hours, ≥135/85 mm Hg for the daytime average, and ≥120/70 mm Hg for the nighttime average (equivalent to office BP >140/90 mm Hg). Sustained hypertension refers to abnormal BP readings in both office and out-of-office settings. The standard echocardiographic parameters were measured following the current guidelines of the European Association of Cardiovascular Imaging.17
The study adhered to the principles specified in the Helsinki Declaration for biomedical research involving human subjects. The study protocol received approval from the Clinical Research Ethics Committee. This study did not utilize any artificial intelligence technology.
Evaluation of Anxiety and HADS Questionnaire
After obtaining BP readings, eligible participants were asked to complete a standardized questionnaire addressing anxiety. This invitation was extended after thoroughly explaining the objectives of the present study and obtaining informed consent. We utilized the HADS-A questionnaire to evaluate anxiety levels in patients with elevated BP in the physician’s office. The HADS-A is a self-assessment questionnaire that can be administered by an interviewer or completed via self-report in the outpatient clinic.18,
The HADS questionnaire consists of 14 items, with 7 dedicated to anxiety (HADS-A) and 7 to depression (HADS-D). In this study, we utilized the HADS-A subscale, which involves items scored on a 4-points Likert scale, ranging from 0 to 3. The total score for the HADS-A subscale ranges from 0 to 21, with a higher score indicating a higher level of anxiety (see
Statistical Analysis
Statistical analyses were performed using R statistical software (version 4.1.3, Vienna, Austria). The normality of variables was assessed using the Kolmogorov–Smirnov test, supported by visual inspections of histograms and probability plots. Continuous variables were presented as mean ± standard deviation for normally distributed data and as median [interquartile range (IQR25-75)] for non-normally distributed data. Categorical data were expressed as numbers and percentages. Group-wise comparisons of categorical variables utilized Fisher’s exact test or the
The relationship between blood pressure at outpatient clinic admission, ABPM records, and HADS-A was assessed through Pearson correlation coefficient analysis. The association between variables and WCH was evaluated through univariate logistic regression analysis. The least absolute shrinkage and section operator (LASSO) penalized selection method was applied to variables identified as significant in univariate logistic regression analysis, utilizing an optimal lambda value for variable reduction to prevent overfitting. Subsequently, nine variables, including HADS-A, remained after this stage. All of them were included in the multiple logistic regression analyses of baseline and full models, incorporating non-linear terms for continuous variables. Odds ratios (OR) and 95% confidence intervals (CI) were calculated for all regression analyses. Multicollinearity was evaluated using the variance inflation factor, where a threshold of >3 indicated significant multicollinearity. The goodness of fit of logistic regressions was assessed using the Hosmer-Lemeshow test.
The model’s performance was assessed using various metrics, including Akaike information criteria (lower values indicating better fit), Brier score (lower values indicating better calibration), Adjusted R2 (higher values indicating better fit), and Harrell’s C-index (higher values indicating better discrimination). To determine individual variable importance within the multiple model, a random-forest-based variable importance method was employed. Model calibration was visually represented through calibration plots, and internal validation was carried out using bootstrap resampling.
The discriminative capability of the full model was compared to the baseline model through pairwise comparisons of receiver-operating characteristics (ROC) curves using the DeLong method. The optimal cut-off value for HADS-A was determined using the Youden index. Additionally, to thoroughly assess the multiple regressions, threshold-performance plots were generated for both the baseline and full models. Decision curve analysis illustrated the net benefit of using the full model over all- and no-treatment strategies, as well as the baseline model, for WCH determination. All statistical analyses employed two-sided tests with a significance level (alpha) of 0.05.
Results
Baseline Characteristics
In this cohort of 303 consecutive participants (mean age 54 years, 46% female), 81 (26.7%) patients were diagnosed with WCH. Individuals with WCH were younger (49 vs. 56 years,
Association of WCH with Anxiety
Correlation analysis indicated that HADS-A was not associated with ABPM parameters in the WCH group. However, it positively correlated with systolic and diastolic BP measured in the outpatient clinic (r = 0.523 and r = 0.387, respectively;
In multiple logistic regression analyses, considering the variables identified by the Lasso penalized selection method, age (OR = 0.937, 95% CI: 0.911-0.965,
Model Performance and HADS-A
The ROC curve analysis demonstrated that the full model, incorporating HADS-A, exhibited superior discriminative ability compared to the baseline model (Harrell’s c-index 0.82 vs. 0.77, respectively,
The ROC curve analysis at a cut-off of >6 for HADS-A revealed a sensitivity of 76.5% and specificity of 53.6% in detecting WCH (area under the curve = 0.72, 95% CI: 0.66-0.79,
Discussion
The key findings of our study include the following: (i) Higher levels of HADS-A in individuals with WCH compared to those with SustHT, (ii) Age, body mass index, heart rate per minute, uric acid, and HADS-A emerged as the most crucial parameters in detecting WCH, with HADS-A showing the strongest association with an increased risk (OR = 1.299), (iii) HADS-A proved to be the parameter contributing the most to the model, alongside heart rate, and this improvement was consistent and sustained, demonstrating a net benefit. To our knowledge, this study is the first to establish a robust analysis demonstrating the relationship between anxiety and WCH.
Sustained hypertension and WCH are prevalent in the general population; however, the detrimental effects on target organs significantly differ between these 2 groups.21,
The autonomic nervous system, particularly its sympathetic arm, plays a crucial role in regulating the cardiovascular system, including BP. Emotional states are believed to exert a profound influence on BP due to this regulatory connection. The relationship between anxiety and hypertension has been extensively investigated, yielding controversial results. While some studies report a positive association between anxiety and hypertension, others do not.32-
The HADS questionnaire is designed to capture the current emotional state of respondents, aiding in the detection and grading of anxiety and depression levels. In its current form, the HADS is divided into four stages: normal (0-7), mild anxiety (8-10), moderate anxiety (11-15), and severe anxiety (16-21).19 The HADS-A score is derived by summing scores for individual items, with scores of 8 or higher indicating clinical anxiety, and a higher score indicating more severe symptoms.
Several studies in the literature have compared the HADS-A questionnaire to measure levels of anxiety in patients with traumatic cerebral lesions, cancers, and rheumatologic diseases, including ankylosing spondylitis, systemic lupus erythematosus, and Sjögren’s syndrome.35-
Notably, to our knowledge, there is no existing study in the literature that investigates the relationship between WCH and HADS-A scores. This metric can offer evidence of generalized symptoms of anxiety and fear, potentially proving valuable in the evaluation of anxiety-related conditions.
Study Limitations
The study has certain limitations that merit acknowledgment. Firstly, the single-center, relatively small sample size, and cross-sectional design inherently limit the ability to establish a cause-and-effect relationship and generalize the findings. Nevertheless, the use of a well-established scale, the HADS-A questionnaire, to explore the connection between anxiety and WCH has significantly contributed to substantiating the hypothesis within the literature. Additionally, our study lacked a control arm of normotensive participants. Lastly, to unveil the interrelationship between anxiety and WCH, further external validation is essential through randomized, multicenter, and expansive participatory studies.
Conclusion
Our study found a heightened level of anxiety, documented by the HADS-A, in patients with WCH compared to SustHT. Identifying WCH is essential for clinicians to differentiate between patients with true hypertension and those with elevated BP readings only in clinical settings. White coat hypertension may be associated with an increased risk of cardiovascular events in certain populations. Recognizing WCH enables clinicians to closely monitor these patients and implement appropriate preventive measures to mitigate long-term cardiovascular risks. Failure to recognize WCH poses the risk of prescribing unnecessary antihypertensive medications to patients who do not have SustHT, leading to unwarranted healthcare costs, potential medication side effects, and patient anxiety. The observed association between WCH and anxiety highlights the importance for clinicians to prioritize the clinical management of WCH while also addressing anxiety treatment goals. Further investigations in this context are warranted to deepen our understanding of these relationships.
Footnotes
References
- White WB, Schulman P, McCabe EJ, Dey HM. Average daily blood pressure, not office blood pressure, determines cardiac function in patients with hypertension. JAMA. 1989;261(6):873-877. https://doi.org/10.1001/jama.1989.03420060089038
- O’Brien E, Parati G, Stergiou G. European Society of Hypertension position paper on ambulatory blood pressure monitoring. J Hypertens. 2013;31(9):1731-1768. https://doi.org/10.1097/HJH.0b013e328363e964
- Franklin SS, Thijs L, Hansen TW, O’brien E, Staessen JA. White-coat hypertension: new insights from recent studies. Hypertension. 2013;62(6):982-987. https://doi.org/10.1161/HYPERTENSIONAHA.113.01275
- Williams B, Mancia G, Spiering W. ESC/ESH Guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur Heart J. 2018;39(33):3021-3104. https://doi.org/10.1093/eurheartj/ehy339
- Cohen JB, Lotito MJ, Trivedi UK, Denker MG, Cohen DL, Townsend RR. Cardiovascular events and mortality in white coat hypertension: a systematic review and meta-analysis. Ann Intern Med. 2019;170(12):853-862. https://doi.org/10.7326/M19-0223
- Abolbashari M. White coat hypertension and cardiovascular diseases: innocent or guilty. Curr Cardiol Rep. 2018;20():1-5.
- Özyüncü N, Çağlar N. Klinikte labil hipertansiyon. Turk Kardiyol Dern Ars. 2019;47(8):701-704. https://doi.org/10.5543/tkda.2019.64009
- Terracciano A, Scuteri A, Strait J. Are personality traits associated with white coat and masked hypertension?. J Hypertens. 2014;32(10):1987-1992. https://doi.org/10.1097/HJH.0000000000000289
- Spruill TM, Pickering TG, Schwartz JE. The impact of perceived hypertension status on anxiety and the white coat effect. Ann Behav Med. 2007;34(1):1-9. https://doi.org/10.1007/BF02879915
- Siegel WC, Blumenthal JA, Divine GW. Physiological, psychological, and behavioral factors and white coat hypertension. Hypertension. 1990;16(2):140-146. https://doi.org/10.1161/01.hyp.16.2.140
- Ogedegbe G. Causal mechanisms of masked hypertension: socio-psychological aspects. Blood Press Monit. 2010;15(2):90-92. https://doi.org/10.1097/MBP.0b013e3283380df5
- Reiff S, Katkin ES, Friedman R. Classical conditioning of the human blood pressure response. Int J Psychophysiol. 1999;34(2):135-145. https://doi.org/10.1016/s0167-8760(99)00071-9
- Taher ZA, Khayyat WW, Balubaid MM, Tashkandi MY, Khayyat HA, Kinsara AJ. The effect of blood pressure variability on the prognosis of hypertensive patients. Anatol J Cardiol. 2019;22(3):112-116. https://doi.org/10.14744/AnatolJCardiol.2019.00905
- Miller NE, Dworkin BR. Effects of learning on visceral functions—biofeedback. N Engl J Med. 1977;296(22):1274-1278. https://doi.org/10.1056/NEJM197706022962210
- Norton S, Cosco T, Doyle F, Done J, Sacker A. The Hospital Anxiety and Depression Scale: a meta confirmatory factor analysis. J Psychosom Res. 2013;74(1):74-81. https://doi.org/10.1016/j.jpsychores.2012.10.010
- Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361-370. https://doi.org/10.1111/j.1600-0447.1983.tb09716.x
- Lang RM, Badano LP, Mor-Avi V. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1-39.e14. https://doi.org/10.1016/j.echo.2014.10.003
- Axford J, Butt A, Heron C. Prevalence of anxiety and depression in osteoarthritis: use of the Hospital Anxiety and Depression Scale as a screening tool. Clin Rheumatol. 2010;29(11):1277-1283. https://doi.org/10.1007/s10067-010-1547-7
- Julian LJ. Measures of anxiety. Arthritis Care Res. 2011;63():-.
- Aydemir Ö, Guvenir T, Kuey L, Kultur S. Validity and reliability of Turkish version of hospital anxiety and depression scale. Turk Psikiyatr Derg. 1997;8():280-287.
- Cai P, Lin Q, Lv D, Zhang J, Wang Y, Wang X. Establishment of a scoring model for the differential diagnosis of white coat hypertension and sustained hypertension. Blood Press Monit. 2023;28(4):185-192. https://doi.org/10.1097/MBP.0000000000000646
- Eguchi K, Hoshide S, Ishikawa J. Cardiovascular prognosis of sustained and white-coat hypertension in patients with type 2 diabetes mellitus. Blood Press Monit. 2008;13(1):15-20. https://doi.org/10.1097/MBP.0b013e3282f13f4a
- Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality. Lancet. 2003;361(9366):1391-1392. https://doi.org/10.1016/S0140-6736(03)13064-4
- Lawes CM, Rodgers A, Bennett DA. Blood pressure and cardiovascular disease in the Asia Pacific region. J Hypertens. 2003;21(4):707-716. https://doi.org/10.1097/00004872-200304000-00013
- Thomas F, Rudnichi A, Bacri AM, Bean K, Guize L, Benetos A. Cardiovascular mortality in hypertensive men according to presence of associated risk factors. Hypertension. 2001;37(5):1256-1261. https://doi.org/10.1161/01.hyp.37.5.1256
- Tully PJ, Harrison NJ, Cheung P, Cosh S. Anxiety and cardiovascular disease risk: a review. Curr Cardiol Rep. 2016;18(12):1-. https://doi.org/10.1007/s11886-016-0800-3
- Cuspidi C, Gherbesi E, Faggiano A, Tadic M. When office blood pressure impacts cardiac mechanics more than ambulatory. Am J Hypertens. 2023;36(1):21-22. https://doi.org/10.1093/ajh/hpac113
- Antza C, Vazakidis P, Doundoulakis I. Masked and white coat hypertension, the double trouble of large arteries: a systematic review and meta‐analysis. J Clin Hypertens (Greenwich). 2020;22(5):802-811. https://doi.org/10.1111/jch.13876
- Pierdomenico SD, Pierdomenico AM, Coccina F, Porreca E. Prognosis of masked and white coat uncontrolled hypertension detected by ambulatory blood pressure monitoring in elderly treated hypertensive patients. Am J Hypertens. 2017;30(11):1106-1111. https://doi.org/10.1093/ajh/hpx104
- Tientcheu D, Ayers C, Das SR. Target organ complications and cardiovascular events associated with masked hypertension and white-coat hypertension: analysis from the Dallas Heart Study. J Am Coll Cardiol. 2015;66(20):2159-2169. https://doi.org/10.1016/j.jacc.2015.09.007
- Bombelli M, Toso E, Peronio M. The Pamela study: main findings and perspectives. Curr Hypertens Rep. 2013;15(3):238-243. https://doi.org/10.1007/s11906-013-0348-1
- Johnson HM. Anxiety and hypertension: is there a link? A literature review of the comorbidity relationship between anxiety and hypertension. Curr Hypertens Rep. 2019;21():1-7.
- Hamam MS, Kunjummen E, Hussain MS, Nasereldin M, Bennett S, Miller J. Anxiety, depression, and pain: considerations in the treatment of patients with uncontrolled hypertension. Curr Hypertens Rep. 2020;22(12):106-. https://doi.org/10.1007/s11906-020-01117-2
- Särnholm J, Kronish IM. Psychological distress and hypertension diagnostic testing: is there anything to worry about?. Am J Hypertens. 2024;37(1):18-20. https://doi.org/10.1093/ajh/hpad096
- Ertenli I, Ozer S, Kiraz S. Infliximab, a TNF-alpha antagonist treatment in patients with ankylosing spondylitis: the impact on depression, anxiety and quality of life level. Rheumatol Int. 2012;32(2):323-330. https://doi.org/10.1007/s00296-010-1616-x
- Mak A, Tang CS-K, Chan M-F, Cheak AA-C, Ho RC-M. Damage accrual, cumulative glucocorticoid dose and depression predict anxiety in patients with systemic lupus erythematosus. Clin Rheumatol. 2011;30(6):795-803. https://doi.org/10.1007/s10067-010-1651-8
- Valtýsdóttir ST, Gudbjörnsson B, Lindqvist U, Hällgren R, Hetta J. Anxiety and depression in patients with primary Sjögren's syndrome. J Rheumatol. 2000;27(1):165-169.
- Pogosova N, Boytsov S, De Bacquer D. Factors associated with anxiety and depressive symptoms in 2775 patients with arterial hypertension and coronary heart disease: results from the COMETA Multicenter Study. Glob Heart. 2021;16(1):73-. https://doi.org/10.5334/gh.1017


